港彩高手出版精料
澳门精华区
香港精华区
- 121期:【贴身侍从】必中双波 已公开
- 121期:【过路友人】一码中特 已公开
- 121期:【熬出头儿】绝杀两肖 已公开
- 121期:【匆匆一见】稳杀5码 已公开
- 121期:【风尘满身】绝杀①尾 已公开
- 121期:【秋冬冗长】禁二合数 已公开
- 121期:【三分酒意】绝杀一头 已公开
- 121期:【最爱自己】必出24码 已公开
- 121期:【猫三狗四】绝杀一段 已公开
- 121期:【白衫学长】绝杀一肖 已公开
- 121期:【满目河山】双波中 已公开
- 121期:【寥若星辰】特码3行 已公开
- 121期:【凡间来客】七尾中特 已公开
- 121期:【川岛出逃】双波中特 已公开
- 121期:【初心依旧】绝杀四肖 已公开
- 121期:【真知灼见】7肖中特 已公开
- 121期:【四虎归山】特码单双 已公开
- 121期:【夜晚归客】八肖选 已公开
- 121期:【夏日奇遇】稳杀二尾 已公开
- 121期:【感慨人生】平特一肖 已公开
- 121期:【回忆往事】男女中特 已公开
- 121期:【疯狂一夜】单双中特 已公开
- 121期:【道士出山】绝杀二肖 已公开
- 121期:【相逢一笑】六肖中特 已公开
- 121期:【两只老虎】绝杀半波 已公开
- 121期:【无地自容】绝杀三肖 已公开
- 121期:【凉亭相遇】六肖中 已公开
- 121期:【我本闲凉】稳杀12码 已公开
- 121期:【兴趣部落】必中波色 已公开
【管家婆一句话】

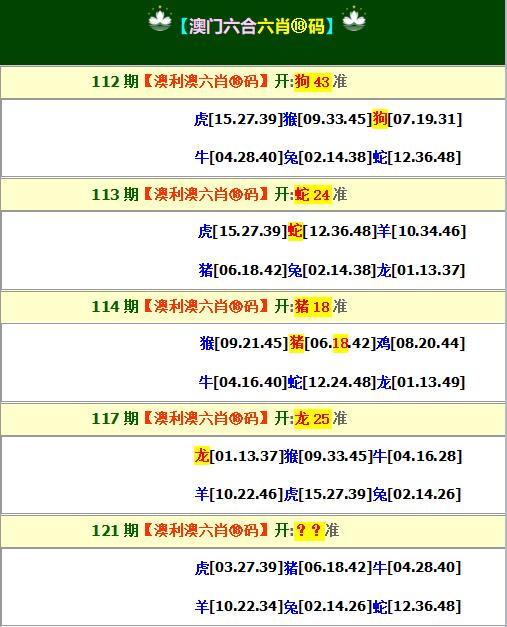
【六肖十八码】
澳门正版资料免费资料大全
- 杀料专区
- 独家资料
- 独家九肖
- 高手九肖
- 澳门六肖
- 澳门三肖
- 云楚官人
- 富奇秦准
- 竹影梅花
- 西门庆料
- 皇帝猛料
- 旺角传真
- 福星金牌
- 官方独家
- 贵宾准料
- 旺角好料
- 发财精料
- 创富好料
- 水果高手
- 澳门中彩
- 澳门来料
- 王中王料
- 六合财神
- 六合皇料
- 葡京赌侠
- 大刀皇料
- 四柱预测
- 东方心经
- 特码玄机
- 小龙人料
- 水果奶奶
- 澳门高手
- 心水资料
- 宝宝高手
- 18点来料
- 澳门好彩
- 刘伯温料
- 官方供料
- 天下精英
- 金明世家
- 澳门官方
- 彩券公司
- 凤凰马经
- 各坛精料
- 特区天顺
- 博发世家
- 高手杀料
- 蓝月亮料
- 十虎权威
- 彩坛至尊
- 传真內幕
- 任我发料
- 澳门赌圣
- 镇坛之宝
- 精料赌圣
- 彩票心水
- 曾氏集团
- 白姐信息
- 曾女士料
- 满堂红网
- 彩票赢家
- 澳门原创
- 黃大仙料
- 原创猛料
- 各坛高手
- 高手猛料
- 外站精料
- 平肖平码
- 澳门彩票
- 马会绝杀
- 金多宝网
- 鬼谷子网
- 管家婆网
- 曾道原创
- 白姐最准
- 赛马会料











